
Six Sigma is a very common manufacturing strategy that is utilised by a large number of manufacturers across various industries. Here at Advant Medical, Six Sigma is one of the methods we use to nurture a lean philosophy and provide innovative manufacturing techniques. We consistently incorporate lean principles that maximise efficiencies, eliminate waste and streamline business processes to effectively increase speed to market for our customers. To learn more about Six Sigma check out our blog here.
There are many methods used within Six Sigma but the two main methodologies are DMAIC and DMADV. Each has its own set of recommended procedures to be implemented for business transformation. This article will focus on explaining the two methodologies, their advantages and their differences and also outline alternative methodologies that are also commonly used. Before comparing the two, let’s first look at the two methodologies in more detail:
DMAIC Methodology
DMAIC Methodology is a data-driven quality strategy used to improve processes. The letters in the acronym represent the five phases that make up the process. The DMAIC improvement cycle is the core tool used to drive Six Sigma projects. Here is an outline of the steps involved in the process:
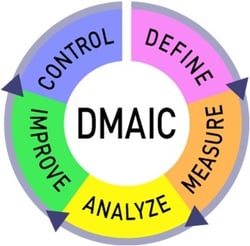
Define
The Six Sigma process begins with a customer-centric approach. In the first phase of the DMAIC process, the project team better defines the problem they have been given by leadership, asking questions to both internal and external customers to affirm that the issue really exists. During this phase, we select the most critical and impactful opportunities for improvement.
Measure
The second phase is focused on the metrics of the project and the tools used in the measurement. This phase aims to ensure that we can measure the problem and understand the current performance of the process before we start trying to improve it. This phase establishes a baseline, which will be particularly useful later in the project when we want to measure the effect of any process improvements made. The ability of a process to meet requirements can be evaluated using capability analysis, and the frequency of issues or their root causes can be examined using a Pareto chart.
Analyse
The analysis phase is all about understanding the root cause of the problem. The goal is to identify and test the underlying causes of problems to make sure that improvement takes place where the problems stem from. One of the biggest challenges many teams face is resisting the urge to jump to a solution before understanding the root causes of process issues. During this phase, the project team collects and uses data to prove theories of root cause or causes of the problem. Crucially the team must verify their hypotheses before implementing solutions. By the conclusion of this phase, the team will have narrowed down their multiple theories to a vital potential few root causes to test and prove true or false. Root cause analysis (RCA) and Failure mode and effects analysis (FMEA) can be used to find causes, as well as Multi-vari charting to identify various types of variation within a process.
Improve
Having established what causes the problem, this phase is where you identify how you can improve the process implementation. With the analysis done and the data in front of you, the project team now begins acting on what they have learned by making improvements. The objective at the end of this stage is to complete a test run of a change that is to be widely implemented.. A structured improvement effort can lead to innovative and elegant changes that improve the baseline measure and, ultimately, the customer experience. Design of experiments (DOE) is a method of problem-solving in complicated processes or systems where numerous variables affect the result and Kaizen event to introduce rapid change by concentrating on a specific project and using the ideas and motivation of the people who do the work.
Control
The last phase is all about ensuring that the new, improved process is stable and under control. The objective of the last stage of the methodology is to develop metrics that help leaders monitor and document continued success. After changes are in place and are successfully addressing the problems to improve your operations, it’s time to bring the process under control to ensure its long-term effectiveness. The project team develops a plan to track the success of the updated process and monitor its effectiveness. The process should continually be updated with current best practices. After the Control phase, you can quantify the complete impact of process changes in terms of cost reduction, efficiency, quality improvement, productivity increase, and customer satisfaction. Statistical process control (SPC) for tracking process behaviour, a quality control plan to outline what is required to maintain an improved process at its current level, and mistake proofing (poka-yoke) to make errors impossible or instantly detectable.
What are the Advantages of Using DMAIC Model?
The main advantages of using the DMAIC model are:
- ● Support for Improvement Culture.
- As a structured approach, DMAIC provides a business with a road map for solutions and is useful in any situation where you need to improve a complex process.
- ● Improved Collaboration.
- It helps to improve team and organisation communications. This leads to improved performance overall, and ultimately this can filter through to happier customers.
- ● More Impact from Improvement.
- DMAIC allows a business to quantify improvements and find answers to complex problems. The DMAIC method aims to analyse a process before implementation, and this reduces the chance of fixing the wrong issue.
- ● Ensure Accuracy.
- DMAIC supports an analytical approach, allowing the business to use the collected data to pinpoint issues and create accurate solutions.
DMADV Methodology
DMADV is a Six Sigma framework that focuses on the development of a new product, service, or process. The DMADV process considers customer requirements at every stage of the creation. When creating high-quality solutions, DMADV keeps the customer in mind. Here is an outline of the steps involved in the process:
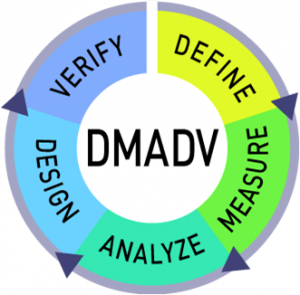
Define
Project goals and client deliverables are identified at this design phase. This step’s goal is to clearly define the project’s problem, goal, scope, resources, and high-level project schedule. Customers are usually thought of as external, but for subassemblies where the manufacturer supplies components to another in-house process, the customer is internal. Indirect sources of information such as industry research and historical data can be used to define what the customer needs. A clear definition of the project is established during this step, and every strategy and goal must be aligned with the expectations of the company and the customers.
Measure
The second step is to use the definition of what the customer wants to create a specification. A specification is a requirement which is clearly stated and defines the product or process in a quantitative way. Steps taken should include defining requirements and market segments, identifying the critical design parameters, designing scorecards that will evaluate the design components more important to the quality, reassessing risk and assessing the production process capability and product capability. It is important here to determine which metrics are critical to the stakeholder and to translate the customer requirements into clear project goals.
Analyse
The proposed process or product is analysed and studied to determine whether there are better ways to achieve the desired results. Actions taken during this phase will include: developing design alternatives, identifying the optimal combination of requirements to achieve value within constraints, developing conceptual designs, evaluating then selecting the best components, and then developing the best possible design. At this stage, you can learn a design’s life cycle cost and set long-term solutions. After thoroughly exploring the different design alternatives, incorporate the ones that improve the process or product.
Design
Based on what was learned in the analysis step, design and test the product, service or process. The elements of the design are prioritised and from there a high-level design is developed. Once this step is complete, a more detailed model will be prototyped to identify where errors may occur and to make necessary modifications. As revisions are made, the analysis step is repeated to compare the new design with the specified characteristics. Customer test groups may be used to test new designs to determine how well they are meeting customer expectations. Within this phase, there are multiple methods used to find the ideal design, such as Design for Manufacturing or Design for Performance.
Verify
Having completed the design, it is essential that the end result meets or exceeds the customer’s requirements. This includes verifying the product is being made as it is supposed to be made, as well as whether it is meeting the requirements of the specification. At this stage the product will be tested, inspect the samples after testing and decide whether or not to scale up the design. During this ongoing process, expectations will be confirmed, deployment will be expanded and all lessons learned will be documented. Even after the product is released, customer feedback should be encouraged and incorporated into future designs. The desired result is to have a product that perfectly meets the customer’s requirements and desires.
What are the Advantages of Using the DMADV Model?
The main advantages of using the DMADV model are:
- ● The process is customer-driven.
- The process aims to achieve maximum customer satisfaction while increasing your profits and minimising your defect rates.
- ● Minimise defect rate.
- The process is designed to reduce defects to no more than 3.4 defects per one million products, where anything not acceptable to your customers is considered a defect.
- ● Focus on the entire production and planning process.
- It focuses on your entire production and planning process, rather than just the final product, and helps you establish areas for improvement before you encounter defects or shortcomings.
DMAIC and DMADV. What’s the difference?
DMADV and DMAIC are two tools adopted for the same mission: achieve Six Sigma performance. DMAIC stands for define, measure, analyse, improve, and control. DMADV stands for define, measure, analyse, design, verify. It is clear that the first three steps and the frameworks are similar. Both approaches are intended to increase the effectiveness and efficiency of a business process. . The two methodologies often cause confusion, and it’s imperative to know what works where. According to Purdue the differences between the two methodologies is as below:
- ● DMAIC addresses the current process; DMADV addresses the design process.
- ● DMAIC reduces/eliminates defects (reactive); DMADV prevents defects (proactive).
- ● DMAIC includes specific solutions; DMADV is part of the solution design process.
- ● DMAIC includes controls to sustain the gains; DMADV includes verification and validation of the finished design.
The below flow chart is designed to assist with the process of selecting the appropriate Six Sigma roadmap:

Image courtesy of Key Differences.
As can be seen from the above chart, the processes are similar but used for different purposes. The best way to remember their difference is that DMAIC is designed to enhance a process, product, or service that the organisation will keep selling, while DMADV works best for launching new features or projects. However, based on the need and scenario that we choose the right tool to apply.
Other Methodologies
Below is a list of some of the other methodologies most commonly used within Six Sigma:
- ● IDOV: Identify, Design, Optimise and Verify
The IDOV methodology (identify, design, optimise, verify) is a component of the Six Sigma management system intended to guide processes involved in creating a new product, service or business process and ensure quality control.
- ● DDICA: Design, Develop, Initialise, Control and Allocate
It is often seen that the tools used for DFSS techniques vary from those used for DMAIC Six Sigma. In particular, DMAIC, DDICA practitioners often use new or existing mechanical drawings and manufacturing process instructions as the originating information to perform their analysis, while DFSS practitioners often use simulations and parametric system design/analysis tools to predict both cost and performance of candidate system architectures.
- ● DMADOV: Define, Measure, Analyse, Design, Optimise and Verify
The only difference between DMADOV and DMADV is the O – for optimise. Optimise involves several product evaluations across multiple performance levels and redesigned if necessary. Further experimentation in DOE occurs here.
- ● DMEDI: Define, Measure, Explore, Develop and Implement
DMEDI methodology focuses on creating processes for a new operation. It’s typically something that organisations do as they become more sophisticated in their use of Six Sigma. It’s the next level – rather than fixing existing processes built without consideration of process improvement, it works to design processes correctly from the outset.
Conclusion
Selecting the correct methodology depends on your process. For example when introducing a new process/Product, it makes sense to use the DMADV method. When you are looking to improve on an existing process the DMAIC method is better suited. However any number of Six Sigma methods will improve a process so it is up to the project team to select the method best suited to their specific issue.
Both DMAIC and DMADV provide a structured process for improvement and when applied properly with the appropriate improvement tools, they can help ensure the success of your projects, which can reduce costs and increase customer satisfaction.
If you want to learn more about what Six Sigma is, its origins and its benefits for medical device manufacturing then check out our blog here.
Advant Medical
Continuous improvement is the heart of new product development. Advant Medical has evolved to become industry experts in medical device product and process development. We’re continually striving to solve problems in better, more customer-focused ways. Our competent team will collaborate with you on your product development journey, customising services to your requirements. Our services revolve around Advant’s robust Quality Management System accredited to ISO 13485:2016. This is why Advant Medical is the trusted ‘Partner of Choice’ for some of the largest MedTech companies in the world.Advant Medical.




